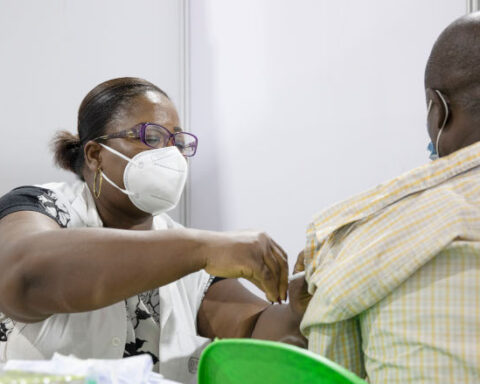
Countries in WHO’s Western Pacific and South-East Asia regions are also affected by these outbreaks. The emergency use listing, or EUL, is the first of its kind for a vaccine and paves the way for potential listing of COVID-19 vaccines.
The world has made incredible progress toward polio eradication, reducing polio cases by 99.9% in the last 30 years. But the last steps to ending this disease are proving the most difficult, particularly with continuing outbreaks of circulating vaccine-derived polio viruses (cVDPVs).
cVDPVs are rare and occur if the weakened strain of the poliovirus contained in the oral polio vaccine (OPV) circulates among under-immunized populations for a long time. If not enough children are immunized against polio, the weakened virus can pass between individuals and over time genetically revert to a form that can cause paralysis. Type 2 cVDPVs are currently the most prevalent form of the vaccine-derived virus.
The EUL procedure and how it could help to speed up access to a future COVID-19 vaccine
The EUL procedure assesses the suitability of yet to be licensed health products during public health emergencies, such as polio and COVID. The objective is to make these medicines, vaccines and diagnostics available faster to address the emergency. The assessment essentially weighs the threat posed by the emergency against the benefit that would accrue from the use of the product based on a robust body of evidence.
The procedure was introduced during the West Africa Ebola outbreak of 2014-2016, when multiple Ebola diagnostics received emergency use listing; since then, numerous COVID-19 diagnostics have also been listed. The nOPV2 is the first such listing for a vaccine.