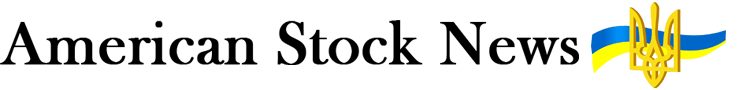The study is taking place in hospitalized adults with COVID-19 in the United States, Mexico and 16 other countries on five continents. The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, is sponsoring and funding the Phase 3 trial, called Inpatient Treatment with Anti-Coronavirus Immunoglobulin, or ITAC.
Trump Receives Dexamethasone, Remdesivir in COVID-19 Hospitalization
Remdesivir is an antiviral drug developed by Gilead Sciences, originally to treat the hepatitis C virus. It did not perform well against that pathogen but has been tried against Ebola and other viruses, after showing some activity in cells and animal models. The drug inhibits a viral enzyme used for replication of the pathogen. Earlier this year, it demonstrated a modest clinical benefit in a trial with hospitalized COVID-19 patients, leading FDA to grant Gilead an EUA for the drug. That EUA has since been expanded for use in patients with mild disease although its benefit in them is not clear. The drug has become widely used for COVID-19 patients despite continuing skepticism that it has a major clinical benefit. Because it and the monoclonal antibodies target different parts of the virus, administering them together may have a synergistic effect. One COVID-19 clinical trial is testing remdesivir and Lilly’s antibody, for example.
The antibody solution being tested in the ITAC trial is anti-coronavirus hyperimmune intravenous immunoglobulin, or hIVIG. The antibodies in anti-coronavirus hIVIG come from the liquid portion of blood, or plasma, donated by healthy people who have recovered from COVID-19. These antibodies are highly purified and concentrated so that the anti-coronavirus hIVIG consistently contains several times more SARS-CoV-2 neutralizing antibodies than typically found in the plasma of people who have recovered from COVID-19.
The ITAC investigators hypothesize that giving people anti-coronavirus hIVIG at the onset of COVID-19 symptoms, before the body makes a protective immune response on its own, could augment the natural antibody response to SARS-CoV-2, thereby reducing the risk of more serious illness and death.
“Finding safe and effective treatments for COVID-19 is absolutely critical,” said NIAID Director Anthony S. Fauci, M.D. “The ITAC trial will examine whether adding anti-coronavirus hIVIG to a remdesivir regimen can give the immune system a needed boost to suppress SARS-CoV-2 early in the course of illness, nipping the infection in the bud.”
Leading the ITAC trial is Protocol Chair Mark Polizzotto, M.D., Ph.D., head of the Therapeutic and Vaccine Research Program at The Kirby Institute in the University of New South Wales, Sydney. The University of Minnesota is the coordinating center for the trial, which is being conducted by the NIAID-funded International Network for Strategic Initiatives in Global HIV Trials (INSIGHT). While INSIGHT was established to conduct clinical studies on HIV, it also has been involved in clinical trials related to influenza-like illness and the role of anti-influenza hIVIG since 2009. The ITAC trial also is known as INSIGHT 013.
Four companies are collaborating to provide anti-coronavirus hIVIG for the trial: Emergent BioSolutions of Gaithersburg, Maryland; Grifols S.A. of Barcelona; CSL Behring of King of Prussia, Pennsylvania; and Takeda Pharmaceuticals of Tokyo. The hIVIG from Emergent BioSolutions and Grifols S.A. was developed with support from the Biomedical Advanced Research and Development Authority, part of the Office of the Assistant Secretary for Preparedness and Response at the U.S. Department of Health and Human Services. CSL Behring and Takeda Pharmaceuticals are providing anti-coronavirus hIVIG on behalf of a partnership of plasma companies called the CoVIg-19 Plasma Alliance.
Remdesivir is currently recommended for treating certain hospitalized patients with COVID-19, based on an analysis of available data from the NIAID-sponsored Adaptive COVID-19 Treatment Trial (ACTT). ACTT found that hospitalized patients with COVID-19 and lower respiratory tract involvement who received remdesivir had a statistically significant shorter time to recovery compared to patients who received placebo. Remdesivir is an investigational broad-spectrum antiviral discovered and developed by Gilead Sciences, Inc. of Foster City, California.
The ITAC study team will enroll 500 hospitalized adults ages 18 or older who provide informed consent, have had COVID-19 symptoms for 12 days or fewer, and do not have life-threatening organ dysfunction or organ failure. >Enrollment will occur at up to 58 sites in Africa, Asia, Europe, North America and South America. Study participants will be assigned at random to receive infusions of either anti-coronavirus hIVIG and remdesivir or a placebo and remdesivir. Neither the participants nor the study team will know who is receiving which treatment regimen.
hIVIG will be given as a single infusion of 400 milligrams (mg) per kilogram of current body weight. Remdesivir infusions will be administered as a 200-mg loading dose followed by a 100-mg once-daily intravenous maintenance dose during hospitalization for up to 10 days in total.
The main goal of the ITAC trial is to compare the health status of participants in the combination treatment group with participants in the remdesivir-only group on day seven. Health status will be based on an ordinal outcome with seven mutually exclusive categories ranging from no limiting symptoms due to COVID-19, to death. These categories capture the full range of severity experienced by hospitalized patients with COVID-19, according to the study investigators.
ITAC study participants will be followed for 28 days. If the trial goes to completion, the primary analysis will be completed after all participants finish 28 days of follow-up.
What’s the evidence for using remdesivir in COVID-19 patients?
Remdesivir is an antiviral drug developed by Gilead Sciences, originally to treat the hepatitis C virus. It did not perform well against that pathogen but has been tried against Ebola and other viruses, after showing some activity in cells and animal models. The drug inhibits a viral enzyme used for replication of the pathogen. Earlier this year, it demonstrated a modest clinical benefit in a trial with hospitalized COVID-19 patients, leading FDA to grant Gilead an EUA for the drug. That EUA has since been expanded for use in patients with mild disease although its benefit in them is not clear. The drug has become widely used for COVID-19 patients despite continuing skepticism that it has a major clinical benefit. Because it and the monoclonal antibodies target different parts of the virus, administering them together may have a synergistic effect. One COVID-19 clinical trial is testing remdesivir and Lilly’s antibody, for example.
A fourth Phase 3 clinical trial evaluating an investigational vaccine for coronavirus disease 2019 (COVID-19) begun enrolling adult volunteers week ago. The trial is designed to evaluate if the investigational Janssen COVID-19 vaccine (JNJ-78436725) can prevent symptomatic COVID-19 after a single dose regimen. Up to 60,000 volunteers will be enrolled in the trial at up to nearly 215 clinical research sites in the United States and internationally.
The Janssen Pharmaceutical Companies of Johnson & Johnson developed the investigational vaccine (also known as Ad.26.COV2.S) and is leading the clinical trial as regulatory sponsor. Janssen, the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, and the Biomedical Advanced Research and Development Authority (BARDA), part of the U.S. Department of Health and Human Services’ Office of the Assistant Secretary for Preparedness and Response, are funding the trial.
U.S. and international trial sites part of the NIAID-supported COVID-19 Prevention Network(link is external) (CoVPN) will participate in the trial. The CoVPN is composed of existing NIAID-supported clinical research networks with infectious disease expertise and designed for rapid and thorough evaluation of vaccine candidates and monoclonal antibodies for the prevention of COVID-19.
“Four COVID-19 vaccine candidates are in Phase 3 clinical testing in the United States just over eight months after SARS-CoV-2 was identified. This is an unprecedented feat for the scientific community made possible by decades of progress in vaccine technology and a coordinated, strategic approach across government, industry and academia,” said NIAID Director Anthony S. Fauci, M.D. “It is likely that multiple COVID-19 vaccine regimens will be required to meet the global need. The Janssen candidate has showed promise in early-stage testing and may be especially useful in controlling the pandemic if shown to be protective after a single dose.”
The Janssen vaccine candidate is a recombinant vector vaccine that uses a human adenovirus to express the SARS-CoV-2 spike protein in cells. Adenoviruses are a group of viruses that cause the common cold. However, the adenovirus vector used in the vaccine candidate has been modified so that it can no longer replicate in humans and cause disease. Janssen uses the same vector in the first dose of its prime-boost vaccine regimen against Ebola virus disease (Ad26.ZEBOV and MVA-BN-Filo) that was recently granted marketing authorization by the European Commission.
Preclinical findings published in Nature(link is external) show that the investigational Janssen COVID-19 vaccine induced neutralizing antibody responses in rhesus macaques and provided complete or near-complete protection against virus infection in the lungs and nose following SARS-CoV-2 challenge. The safety, reactogenicity and immunogenicity of the investigational vaccine are being evaluated in a Phase 1/2a trial in the United States and Belgium enrolling adult volunteers. Positive interim results from the Phase 1/2a clinical study demonstrated that the safety profile and immunogenicity after a single vaccination were supportive of further development.
“Scientific partners from government, industry and academia are working hand-in-hand to develop safe, effective vaccines to put this pandemic in our rear-view mirror,” said NIH Director Francis S. Collins, M.D., Ph.D. “While administrative steps are being streamlined to speed the process, safety and effectiveness measures are just as rigorous than ever.”
The Phase 3 trial is being conducted in collaboration with Operation Warp Speed(link is external) (OWS), a multi-agency collaboration overseen by HHS and the Department of Defense that aims to accelerate the development, manufacturing and distribution of medical countermeasures for COVID-19. OWS and CoVPN also are assisting with additional COVID-19 preventive candidate vaccines, including mRNA-1273, an investigational vaccine co-developed by NIAID and the Cambridge, Massachusetts-based biotechnology company Moderna, Inc., and AZD1222, a vaccine candidate being developed by United Kingdom-based biopharmaceutical company AstraZeneca.
Source: The FINANCIAL