The United States will soon reopen its air borders for foreign visitors who are fully vaccinated with an approved vaccine or those who can present a negative COVID-19 test within 24 hours of travel, the White House announced Monday.
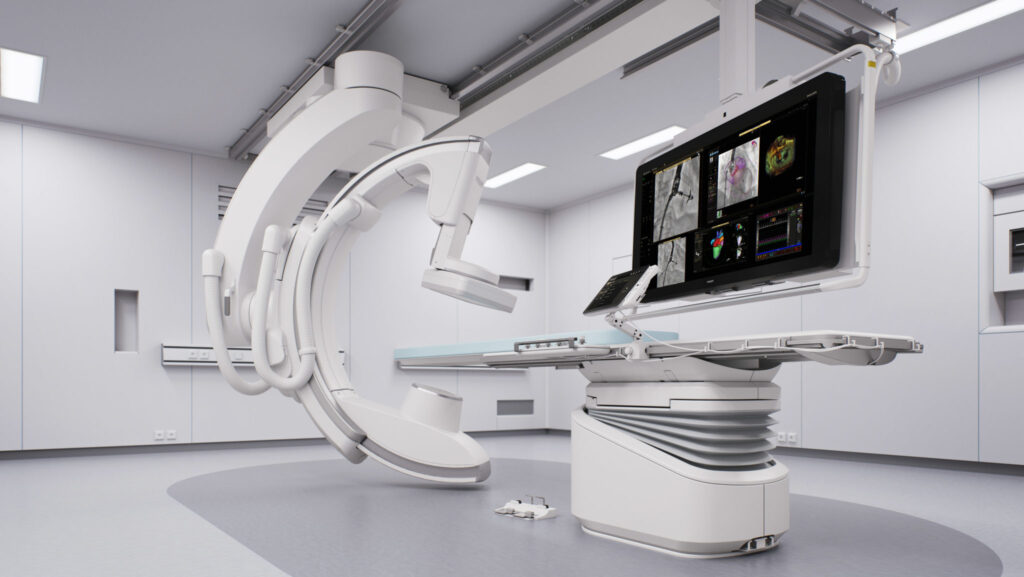
MoreRoyal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced U.S. Food and Drug Administration 510 (k) clearance for its Philips SmartCT application software.
MoreThe Elecsys® SARS-CoV-2 Antigen test is an automated laboratory assay intended as an aid in the diagnosis of SARS-CoV-2 infection
MoreIn a continuing effort to provide the facts about ID NOW to support public health interests, Abbott (NYSE: ABT) is sharing new interim clinical data results on its ID NOW COVID-19 rapid test. The results confirm the data submitted to the U.S. Food and Drug Administration (FDA) in March for Emergency Use Authorization (EUA) and
More